Structural biologist Dr Thomas Ve from the Institute for Glycomics has been awarded an Australian Research Council Future Fellowship for his research into the biology of enzymes involved in protecting bacteria against viral infection and destruction of nerve fibres (axons) in humans.
“These enzymes use an essential helper molecule found in all living cells called nicotinamide adenine dinucleotide (NAD+) as fuel, which sets off a series of events in the cell that can result in its self-destruction,” Dr Ve said.
Dr Ve’s team will use structural biology methods combined with cell and chemistry-based approaches through national and international collaborators to characterise these NAD+ consuming enzymes at the molecular level; define how the enzymes are regulated; and explore the diversity and targets of the nucleotide signals produced by the enzymes.
One member of this enzyme family, SARM1 is a key executioner of axon degeneration.
“In healthy nerve cells, SARM1 is present but inactive. However, disease and injury can trigger SARM1 activation resulting in very rapid breakdown of NAD+, which ultimately leads to destruction of the axon,’’ Dr Ve said.
“Axon loss is common in some of the most prevalent neurological diseases, including peripheral neuropathies, ALS, MS, traumatic brain injury, Parkinson’s disease and glaucoma.
“SARM1 is a potential therapeutic target for these neurodegenerative diseases but to exploit the full promise of targeting SARM1 we must understand the molecular mechanisms of how SARM1 is regulated, activated and cleave NAD+.”
Several other members of this enzyme family play an important role in bacterial defence systems against viruses, but the mechanism of how they use NAD+ to provide resistance against viral infection has not yet been explored.
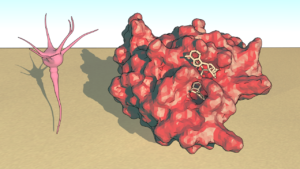
Brain cell and three-dimensional structure of SARM1 modelled with nicotinamide adenine dinucleotide. Credit: Hayden Burdett
Dr Ve said that mechanistic understanding of bacterial antiviral defence systems has previously led to the development of revolutionary biotechnological tools such as restriction enzymes and the genome -editing technology CRISPR-Cas9.
“Understanding the mechanism of new defence systems such as the ones using NAD+ as fuel may provide a foundation for developing new exciting molecular tools.”
